Thirty Years After Angel, the Bioelectric Pieces Are Finally Aligning
By John Coates, Founder of RF Safe
In 1995, my daughter Angel Leigh Coates came into this world and left it almost immediately. She died from a catastrophic neural tube defect. There are no words that make that kind of loss smaller. There is only the question that remains afterward: why?
Two years later, in 1997, a paper appeared in Bioelectromagnetics that changed the direction of my life: “The effect of pulsed and sinusoidal magnetic fields on the morphology of developing chick embryos” by Farrell, Litovitz, Penafiel, Montrose, Doinov, Barber, Brown, and Litovitz. The study did not prove what caused Angel’s condition. Science does not work that way. But it did something that was, for me, impossible to dismiss: it showed that weak, time-varying electromagnetic fields could disturb early embryonic development in a vertebrate model. That paper became one of the sparks behind RF Safe, which I founded in 1998 after years of asking how invisible electromagnetic exposures might interact with the earliest stages of life.
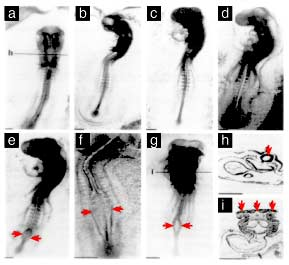
The image at the top of this post is hard to look at. It should be. It shows what the abstract language of “morphological abnormalities” really means: development interrupted. Patterning disrupted. A living system that was supposed to fold, close, differentiate, and organize itself in a precise sequence somehow losing fidelity.
For nearly thirty years, I have carried that picture in my mind. And for nearly thirty years, the dominant regulatory answer has been almost insultingly simple: if it does not heat tissue, it does not matter.
That answer was never good enough. Today, it is scientifically obsolete.
What the 1997 chick-embryo study actually found
The Farrell/Litovitz study was not a casual experiment. It reported experiments conducted over five years, in five separate campaigns, involving more than 2,500 White Leghorn chick embryos. Four of the campaigns used pulsed magnetic fields; the final campaign used a 60 Hz sinusoidal magnetic field. When the campaigns were analyzed separately, four showed statistically significant increases in abnormality rates, ranging from about two-fold to seven-fold. When all pulsed-field campaigns were pooled, pulsed magnetic-field exposure approximately doubled the abnormality rate. The 60 Hz sinusoidal exposure approximately tripled it.
One campaign did not show a statistically significant response. That detail matters. It is not a weakness to hide; it is a clue. The authors reported that the genetic composition of the breeding stock had been changed before the non-responding campaign and hypothesized that genetic composition could determine flock susceptibility to EMF-induced abnormalities. In other words, even in 1997, the study was pointing toward what we would now call a gene-environment or susceptibility-window problem, not a crude one-size-fits-all toxicology model.
That was the first piece of the puzzle for me.
Not “EMF causes every defect.” Not “every embryo responds the same way.” Not “all fields are equally dangerous.”
The real message was more precise and more unsettling:
Early development may contain sensitive biological timing systems that can be perturbed by weak, patterned electromagnetic fields, and susceptibility may depend on biological state, genetics, and timing.
That is exactly the kind of question our federal safety system should have investigated aggressively. Instead, the public was given a thermal standard.
Why chick embryos mattered
A chick embryo is not a human fetus. It is not proof of human causation. But chick embryos have long been important in developmental biology because early vertebrate development is a process of organized pattern formation: folding, closure, migration, segmentation, differentiation, and electrical/chemical signaling.
The reason the 1997 study mattered so deeply is that the earliest embryo is not merely “growing.” It is executing a timed developmental program. During neurulation, the embryo must coordinate cell shape changes, neural-fold elevation, neural-tube closure, and tissue patterning. Timing is everything.
This is where modern developmental biology has caught up with what that 1997 paper was hinting at. We now know that bioelectric signaling is not a fringe concept. Endogenous ion flows, membrane voltages, voltage gradients, calcium transients, and electric fields help guide cell behavior during development and regeneration. A classic chick-embryo study by Hotary and Robinson directly tested whether endogenous electrical fields play a causal role in chick development. Modern reviews now describe bioelectric signaling as part of the information architecture of development, working alongside genes and biochemical gradients.
Calcium is especially important. Reviews of vertebrate development describe calcium signaling as involved in neural induction, early neurogenesis, neural progenitor differentiation, and neural-tube closure. Other work has shown that T-type calcium channels can regulate neural-tube closure pathways in chordate models.
This is the point that was missing from the old regulatory mindset:
The embryo is not just vulnerable to heat. It is vulnerable to mistimed information.
The wrong question: “Does it heat?”
The FCC’s RF exposure framework has historically centered on specific absorption rate, power density, and time-averaged heating. That may be relevant for preventing acute thermal injury. But the question raised by the chick-embryo evidence was never mainly about cooking tissue.
The question was whether weak, patterned electromagnetic fields can perturb developmental signaling.
That distinction matters because biological systems do not operate only by bulk energy deposition. They operate through timing, gradients, ion flux, charge separation, calcium waves, voltage-gated channels, mitochondrial redox state, and gene-expression timing. A field can be too weak to heat tissue and still be relevant if it perturbs the timing of a sensitive biological switch.
That is why the 1997 study stayed with me. The embryos were not being microwaved. The fields were weak. The reported effects were developmental. The suspected sensitivity was not uniform. That is the profile of a nonlinear biological signaling problem, not a simple thermal toxicity problem.
The second piece: timing, pulsing, and coherence
Farrell and Litovitz did not just raise the issue of weak fields. Their broader research program also pointed toward timing and coherence. Later work from the same research lineage reported that a temporally incoherent magnetic “noise” field could inhibit 60 Hz-induced changes in ornithine decarboxylase activity in developing chick embryos. Another related study reported a dose-response relationship for electromagnetic-field-enhanced ornithine decarboxylase activity in L929 cells, with a sigmoidal response and a 50% point around 5 microtesla.
That does not prove a universal mechanism. Replication controversies existed, and some attempts failed to reproduce ODC effects. A 1999 replication effort reported that experiments using key elements of the original exposure system did not demonstrate ODC enhancement.
But the mixed record does not erase the core lesson. It refines it.
Biological EMF responses, when they occur, may depend on waveform, timing, coherence, biological state, developmental stage, and experimental conditions. That means a serious safety program must characterize the signal, the target tissue, and the timing window. It cannot simply average power over minutes and declare the question closed.
Why this matters even though wireless radiation is RF, not 60 Hz magnetic field
A reasonable critic might say: the 1997 chick-embryo study involved pulsed and sinusoidal magnetic fields, not modern Wi-Fi, Bluetooth, LTE, or 5G.
That is correct. And it is exactly why the question must be investigated, not dismissed.
Modern wireless signals are not simple continuous waves. A GHz carrier may be the nominal frequency, but real wireless systems also contain timing structure: modulation, packet intervals, duty cycles, pulse repetition, beaconing, frame structures, beamforming patterns, and low-frequency envelopes. The biological question is not whether a 2.4 GHz carrier is the same thing as a 60 Hz magnetic field. It is whether the low-frequency temporal structure riding on modern wireless emissions can interact with biological timing systems that operate through ion channels, calcium oscillations, and mitochondrial redox rhythms.
A 2025 review by Panagopoulos and colleagues argues that anthropogenic wireless fields differ from simple natural background fields because they are polarized, coherent, modulated, pulsed, and variable, and it proposes that voltage-gated ion-channel dysfunction can trigger calcium disturbance, mitochondrial effects, nitric oxide synthase effects, reactive oxygen species, and oxidative stress. That proposed mechanism should not be treated as final proof. But it directly addresses the question regulators avoided: how could weak, non-thermal fields matter biologically?
This is the bridge from 1997 to today.
In 1997, we saw developmental morphology disrupted in a weak-field model.
In 2025 and 2026, we are finally getting candidate molecular pathways that could explain how patterned fields might disturb biological timing.
The S4 voltage sensor: the nanoscale gate
One of the most important modern pieces is the S4 voltage sensor in voltage-gated ion channels.
Voltage-gated calcium, sodium, and potassium channels are not metaphors. They are molecular machines that control electrical excitability, calcium entry, neurotransmission, muscle contraction, development, and gene regulation. Their S4 segments contain positively charged residues that help sense voltage and control gating.
The modern S4/VGIC hypothesis is not that a weak external field rips open a cell membrane. It is subtler: a slow-varying or pulsed field may influence the motion of ions in or near the channel pore, and because those ions are extremely close to the S4 voltage sensor, even very small displacements may alter gating probability in a nonlinear way. That is why geometry matters. Nanometer-scale spacing changes the force problem.
This is where the old “too weak to matter” objection becomes experimentally testable. If the relevant biological variable is nanoscale ion-channel timing, then average whole-body heating is the wrong measurement. The question becomes: what waveform, what pulse structure, what field orientation, what tissue geometry, what developmental stage, and what channel state?
That is not a claim of settled harm. It is a research demand.
The FDA has already acknowledged non-thermal RF calcium-channel biology as regulatorily relevant
One of the clearest real-world precedents is the FDA-authorized TheraBionic P1 device.
FDA identifies TheraBionic P1 as a handheld RF electromagnetic-field generator used for adults with advanced hepatocellular carcinoma who have failed first- and second-line therapy. FDA says the antenna is placed in the mouth and emits electromagnetic fields at specific amplitude-modulated frequencies. FDA’s public page also says the device should not be used in people receiving calcium channel blockers.
The FDA Summary of Safety and Probable Benefit identifies the device as an Amplitude-Modulated Radiofrequency Electromagnetic Fields device, gives the HDE number H220001, and states that the device is indicated for adults with advanced hepatocellular carcinoma who fail first- and second-line therapy. Its contraindications include patients receiving calcium channel blockers or agents blocking L-type or T-type voltage-gated calcium channels unless treatment is modified to exclude those blockers.
That matters. FDA did not authorize TheraBionic because it heats tissue. The record describes a non-thermal, amplitude-modulated RF medical device with calcium-channel relevance. The FDA pathway was a Humanitarian Device Exemption, which is a probable-benefit pathway, not the same as a full PMA effectiveness standard. That caveat should be stated clearly. But the regulatory point remains powerful: non-thermal RF effects involving calcium-channel biology are not impossible, imaginary, or irrelevant. They are already part of an FDA-regulated medical-device record.
The 1997 chick-embryo study asked whether weak, patterned fields could disturb development. TheraBionic shows that federal regulators can recognize biologically meaningful, non-thermal, amplitude-modulated RF interactions with calcium-channel biology in a clinical setting.
The CYB5B discovery: a candidate mitochondrial EMF sensor
The newest and most important piece may be CYB5B.
In 2026, Kim and colleagues published a Cell paper describing an electromagnetic-field-inducible in vivo gene switch. According to PubMed, the authors used a CRISPR-Cas9 screen and identified cytochrome b5 type B, CYB5B, as an essential mediator likely acting as an EMF sensor. The abstract also states that activation depended on rhythmic oscillatory calcium dynamics rather than generic calcium influx.
That distinction is everything.
The question is not simply whether calcium goes up or down. The question is whether the calcium waveform remains faithful: frequency, amplitude, phase, burst timing, recovery kinetics, and spatial localization. In a developing embryo, those patterns can mean the difference between proliferation and differentiation, closure and failure, organization and disorganization.
This does not prove that everyday Wi-Fi or cell towers activate CYB5B in harmful ways. It does something narrower and stronger: it gives researchers a place to look. Before CYB5B, agencies could say that non-thermal weak-field biology lacked a concrete molecular transducer. After CYB5B, the question is falsifiable.
Does wireless-relevant pulsing or modulation affect CYB5B-mediated calcium oscillation? Does CYB5B manipulation alter the biological response? Does CYB5B status change developmental vulnerability? Does it interact with S4/VGIC signaling? These are experiments that can be done.
The BET organoid study: development reads timing
The next piece is the 2025 Cell Reports study by Cakir and colleagues: “Radiofrequency regulates the BET-mediated pathways in radial glia differentiation in human cortical development.”
Using human cortical organoids derived from human embryonic stem cells, the authors investigated RF effects on corticogenesis. The abstract reports that RF exposure regulated differentiation of human and non-human primate radial glia progenitors, maintained stem-cell identity, delayed differentiation, induced expression of human endogenous retroviruses in differentiated neurons, and that BET inhibitors rescued RF-induced developmental defects in human cortical organoids.
The exposure model is directly relevant to modern wireless questions. The study used a 2.4 GHz Bluetooth module operating at 4 dBm, about 2.4 mW, and reported a maximum measured field strength of 2.5 mW/m². The authors used methods including immunohistochemistry, single-cell RNA sequencing, single-cell ATAC sequencing, patch clamp electrophysiology, calcium imaging, and qPCR.
This study should be framed carefully. It does not prove that ordinary wireless exposure causes autism or any specific human neurodevelopmental disorder. Organoids are models, not whole human beings. But it is directly relevant to the question regulators have been avoiding: can RF exposure alter human developmental signaling without relying on tissue heating?
The answer from this model is: yes, at least under the tested conditions, RF exposure altered developmental programs, calcium/electrophysiological behavior, chromatin accessibility, and BET-mediated pathways.
This is where the pieces finally begin to align.
Bioelectric Dissonance: not wireless gene therapy, but corrupted timing
The concern is not that environmental RF acts like “wireless gene therapy” rewriting DNA. That is the wrong frame.
The concern is that pulsed or modulated electromagnetic exposure may degrade the timing fidelity of the calcium-redox signals that tell cells when and how to read the genome.
That is what I mean by Bioelectric Dissonance.
A healthy developing cell does not operate on one constant calcium level. It uses calcium rhythms. It uses membrane voltage. It uses mitochondrial redox state. It uses oscillations, gradients, and timing patterns. These are not side effects of life; they are part of the operating system of life.
Under the hypothesis now worth testing, the chain looks like this:
Pulsed or modulated EMF exposure
→ S4/VGIC and/or CYB5B calcium-timing perturbation
→ loss of calcium waveform fidelity
→ altered mitochondrial redox and kinase/phosphatase signaling
→ altered chromatin acetylation and accessibility
→ mistimed BET protein recruitment
→ radial glia self-renewal/differentiation imbalance
→ altered neuronal maturation and network activity
That is a proposed adverse outcome pathway, not a settled disease claim.
But it is no longer vague. It is testable.
Why the 1997 genetic-susceptibility clue now looks prophetic
One of the most important parts of the Farrell/Litovitz paper was not only the positive result. It was the non-response campaign.
The authors did not hide it. They tried to explain it. Their hypothesis was that the genetic composition of the breeding stock might determine susceptibility to EMF-induced abnormalities.
That idea was ahead of its time.
Today, toxicology increasingly recognizes that vulnerability depends on biological context: genetics, developmental stage, tissue type, mitochondrial density, redox state, ion-channel expression, immune status, and timing of exposure. In our FCC comments, we have described this as density gating: the idea that susceptibility may be highest in tissues with dense excitable membranes, dense voltage-gated channels, dense mitochondrial machinery, intense calcium flux, and high redox demand.
The chick embryo is exactly the kind of system where density gating and developmental timing would matter. Early embryonic tissues are building form through precisely timed signals. They do not have the redundancy of a mature adult system. A small timing disruption at the wrong window can become a large patterning error.
That is why the 1997 study never stopped mattering.
It was not just a result. It was a map of the kind of biology regulators were not measuring.
The prenatal RF animal evidence closes part of the loop
The chick-embryo study raised the developmental question. Later animal studies kept that question alive.
In 2012, Aldad, Gan, Gao, and Taylor published a Scientific Reports mouse study on fetal radiofrequency exposure from 800–1900 MHz-rated cellular telephones. The authors reported that mice exposed in utero were hyperactive and had impaired memory as adults, and that patch-clamp recordings showed altered glutamatergic synaptic transmission in layer V pyramidal neurons of the prefrontal cortex. The authors called for further experiments in humans or non-human primates to determine pregnancy-related risk.
This study also must be framed carefully. Mice are not humans. A cell phone above a cage is not the same as every human exposure condition. But it is developmental, prenatal, neurophysiological, and behavioral. It belongs in the same category of evidence as the 1997 chick-embryo work and the 2025 human cortical organoid study: evidence that developmental windows must be studied directly.
That is what the FCC’s thermal framework does not do.
What the 1997 study did not prove
For credibility, this must be said plainly.
The 1997 chick-embryo study did not prove that EMF caused my daughter’s neural tube defect.
It did not prove that all electromagnetic fields are harmful.
It did not prove that every embryo is equally susceptible.
It did not prove that modern cell phones, Wi-Fi, or 5G reproduce the same exposure conditions.
It did not prove a complete human disease pathway.
But it did prove enough to demand serious investigation. It showed that weak, time-varying electromagnetic fields could be associated with increased developmental abnormalities in a large vertebrate embryo study, and that susceptibility might depend on genetics or biological state.
That is more than enough to reject the lazy claim that non-thermal developmental effects are impossible.
What we should have done after 1997
After the Farrell/Litovitz paper, the obvious federal research program should have been:
- replicate the chick-embryo findings with rigorous exposure characterization;
- identify whether abnormalities were neural-tube, cardiac, vascular, or generalized patterning defects;
- test waveform differences: sinusoidal, pulsed, modulated, continuous, coherent, incoherent;
- measure calcium signaling, ODC activity, oxidative stress, membrane voltage, mitochondrial redox state, and developmental gene expression;
- test genetic susceptibility across strains;
- evaluate whether RF carriers with low-frequency modulation envelopes produce similar developmental endpoints;
- translate the work into mammalian prenatal models and, eventually, human developmental organoids.
Instead, for decades, the public-health conversation was forced back into the same narrow question: does it heat tissue?
That was the wrong question.
What we can do now
Today, we can finally design the experiment the 1997 study deserved.
A modern version would not simply count malformed embryos. It would measure the timing systems that control morphology.
A serious 2026–2027 research program should combine:
- chick embryos, mouse prenatal models, and human cortical organoids;
- sham-controlled and blinded exposure systems;
- continuous-wave versus pulsed/modulated exposures at the same average power;
- wireless-relevant waveforms, including Bluetooth-like, Wi-Fi-like, GSM-like, LTE/5G-like, and TheraBionic-like comparator protocols;
- S4/VGIC perturbation using channel blockers and electrophysiology;
- CYB5B knockdown, knockout, rescue, and mutant rescue;
- calcium waveform imaging, not just total calcium measurement;
- mitochondrial redox endpoints, including NADH/NAD+, FAD/FADH2, ROS, nitric oxide, peroxynitrite, glutathione, and membrane potential;
- chromatin accessibility, histone acetylation, BET occupancy, and single-cell RNA sequencing;
- developmental endpoints: neural-tube closure, radial glia differentiation, neuronal maturation, synapse formation, and network activity.
The decisive question is simple:
If CYB5B or S4/VGIC pathways are upstream of RF-induced developmental changes, then altering those pathways should alter the response.
If CYB5B knockdown blocks an RF-induced calcium/BET phenotype, that supports CYB5B necessity. If wild-type CYB5B rescue restores the response, that supports specificity. If heme-binding or electron-transfer mutants fail to restore the response, that identifies which CYB5B function matters. If the RF phenotype persists unchanged without CYB5B, then the hypothesis is wrong or incomplete.
That is what real science looks like.
Why Public Law 90-602 matters
This is not only a scientific issue. It is a legal and public-health issue.
Congress already gave HHS/FDA authority over electronic product radiation. FDA’s summary of the Electronic Product Radiation Control provisions states that electronic product radiation includes ionizing or non-ionizing electromagnetic radiation emitted from electronic products as a result of electronic circuits. FDA’s examples include cordless and cellular telephones.
21 U.S.C. § 360ii requires the Secretary to establish and carry out an electronic product radiation control program designed to protect public health and safety, including research, exposure evaluation, and procedures for minimizing unnecessary electronic product radiation.
That law was not written only for X-rays. It was written because Congress understood that electronic products can emit radiation and that public-health agencies must study, measure, and control unnecessary exposure.
The question today is whether our agencies will finally apply that mandate to modern wireless exposure in a biologically realistic way.
The promise behind RF Safe
RF Safe was not founded as a product idea. It was founded as a promise.
A promise made in grief.
A promise to ask the questions that were not being asked.
A promise to challenge the assumption that invisible exposure is harmless just because it is convenient.
A promise to protect children, pregnant mothers, and developing life from unnecessary radiation exposure while the science is still being resolved.
The 1997 chick-embryo paper gave that promise a scientific direction. It showed that weak electromagnetic fields could disturb development in a sensitive model. It suggested that genetics and biological state could determine susceptibility. It raised the possibility that the earliest stages of life are governed by bioelectric timing systems that our safety standards were never designed to protect.
Now, almost thirty years later, the pieces are finally visible.
Developmental bioelectricity tells us that embryos use ion flows, voltages, and calcium signals to build form.
The S4/VGIC model gives us a nanoscale voltage-sensor mechanism to test.
TheraBionic P1 gives us an FDA-recognized non-thermal, amplitude-modulated RF calcium-channel precedent.
CYB5B gives us a candidate mitochondrial EMF-responsive mediator.
The Cakir human cortical organoid study gives us a developmental model where RF exposure altered radial glia differentiation and BET-mediated pathways.
Aldad gives us prenatal mammalian evidence that fetal RF exposure can alter adult neurobehavior and prefrontal synaptic physiology.
Together, these do not prove every claim. They do something better: they define the experiments that must be done.
The final lesson of the chick embryos
The lesson of the 1997 study is not fear. It is fidelity.
Life begins as a timed pattern. Cells must receive the right signals at the right moments. Neural folds must rise and close. Calcium must pulse in proper rhythm. Mitochondria must maintain redox balance. Chromatin must open and close at the right developmental windows. BET proteins must read the correct acetylation landscape. The embryo must build itself from signals.
If artificial electromagnetic timing structures can degrade those signals, then the harm is not best understood as heat.
It is Bioelectric Dissonance: the loss of timing fidelity in the living systems that build and maintain life.
That is why the chick-embryo study still matters.
That is why RF Safe exists.
And that is why, after thirty years, I believe the world finally has the tools to answer the question that began with Angel:
Are we measuring the right thing when we claim wireless exposure is safe?
Until HHS, FDA, FCC, and independent researchers answer that question using modern developmental biology, modern waveform science, and modern molecular tools, the only responsible path is precaution, transparency, and aggressive investigation.
The embryos in that image were not just abnormal data points.
They were an early warning that development is electrical, timing matters, and life can be disrupted long before tissue ever heats.
References and studies discussed
Farrell JM, Litovitz TL, Penafiel M, Montrose CJ, Doinov P, Barber M, Brown KM, Litovitz TA. The effect of pulsed and sinusoidal magnetic fields on the morphology of developing chick embryos. Bioelectromagnetics. 1997;18(6):431–438.
IARC/NCBI Bookshelf summary of ELF magnetic-field developmental evidence, including Farrell et al. 1997 chick-embryo findings.
Farrell JM, Barber M, Krause D, Litovitz TA. The superposition of a temporally incoherent magnetic field inhibits 60 Hz-induced changes in the ODC activity of developing chick embryos. Bioelectromagnetics. 1998.
Mullins JM, Penafiel LM, Juutilainen J, Litovitz TA. Dose-response of electromagnetic field-enhanced ornithine decarboxylase activity. 1999.
Cakir B, Tanaka Y, Choe MS, et al. Radiofrequency regulates the BET-mediated pathways in radial glia differentiation in human cortical development. Cell Reports. 2025.
Kim J, Hwang Y, Kim S, et al. Electromagnetic field-inducible in vivo gene switch for remote spatiotemporal control of gene expression. Cell. 2026.
FDA. TheraBionic P1 – H220001. Recently Approved Devices.
FDA. HDE H220001: Summary of Safety and Probable Benefit — TheraBionic P1.
Aldad TS, Gan G, Gao XB, Taylor HS. Fetal Radiofrequency Radiation Exposure From 800–1900 MHz-Rated Cellular Telephones Affects Neurodevelopment and Behavior in Mice. Scientific Reports. 2012.
Hotary KB, Robinson KR. Evidence of a role for endogenous electrical fields in chick embryo development. Development. 1992.
Paudel S, et al. Review of calcium signaling in vertebrate development and neural-tube closure.
Abdul-Wajid S, et al. T-type calcium channel regulation of neural tube closure and EphrinA/EPHA expression.
FDA. Summary of Electronic Product Radiation Control Provisions of the Federal Food, Drug, and Cosmetic Act.
21 U.S.C. § 360ii. Electronic Product Radiation Control program of control.







