From a 60 Hz Gene Switch to an FDA-Approved RF Cancer Device:
For years, the EMF debate was boxed into a crude binary: either a field heated tissue enough to matter, or it was biologically irrelevant. That framing is now under real strain. A 2026 Cell paper showed that a precisely defined electromagnetic-field program can be converted into a distinctive calcium oscillation pattern that remotely controls gene expression in vivo, while the FDA-approved TheraBionic P1 device shows that non-thermal, amplitude-modulated radiofrequency fields can be clinically relevant and are important enough biologically that the FDA labeling specifically warns against concurrent use of calcium-channel blockers. These are not the same system, not the same waveform, and not the same channel subtype. But together they make one thing clear: calcium-channel-dependent EMF biology can no longer be dismissed as impossible merely because it is non-thermal.
The new Cell paper is the sharper mechanistic advance. Cell’s indexed abstract and summary describe an electromagnetic-field-inducible in vivo gene switch for remote spatiotemporal control of gene expression. Search-indexed full-text snippets say the authors identified a 450-bp upstream sequence, designated the EMF-inducible DNA element (Ei), and that their EMF system generated unipolar pulsed fields in which each 16.67 ms cycle at 60 Hz contained a 5 ms active burst of 20 consecutive single pulses, followed by 11.67 ms of silence. Indexed reporting on the paper describes 2.0 mT at 60 Hz as the optimized condition for the Lgr4-derived element. A CRISPR-Cas9 screen identified Cyb5b as an essential upstream mediator, and Cell’s own indexing states that Cyb5b and Cacna1f are required for the distinctive EMF-specific calcium oscillatory pattern that activates the switch.
That mechanistic detail matters because Cacna1f is the gene for CaV1.4, an L-type voltage-gated calcium-channel alpha subunit that forms the pore through which calcium enters the cell. In other words, the Cell paper is not saying CYB5B is itself the calcium pore. It is saying that CYB5B sits upstream as the EMF-responsive mediator, while the actual calcium-flow step runs through a voltage-gated calcium channel. That is the right way to frame the biology.
The most important conceptual point in the Cell paper is not merely that calcium went up. It is that a specific oscillatory pattern mattered. That fits the broader calcium-signaling literature. Calcium signals are not just “more” or “less”; cells decode timing, frequency, duty cycle, and waveform structure. Classic work showed that calcium oscillations can increase the efficacy and information content of calcium-dependent gene expression, and frequency decoding models show that different downstream pathways can respond differently even when average calcium exposure is similar.
That is why this paper should be read as a study of signal writing, not merely stimulation. In the Tph2 arm of the paper, Cell’s indexed text shows that the authors explicitly compared cyclic EMF with continuous EMF in vivo. That alone tells you they were treating temporal patterning as a design variable, not a nuisance parameter. The paper summary also says the Ei platform was used to model Alzheimer’s disease and restore serotonergic function in vivo, which makes the calcium-rhythm question central rather than incidental.
TheraBionic P1 expands the conversation in a different direction. FDA’s HDE materials say the device is approved for adults with advanced hepatocellular carcinoma who have failed first- and second-line therapy. The SSPB describes a 27.12 MHz carrier, amplitude-modulated at tumor-specific frequencies within a 0.01 Hz to 150 kHz range. The same FDA document says the amount of EMF delivered is estimated to be 100 to 1000 times lower than that delivered by cellular phones and does not result in thermal heating in the brain or other specific organs. Most importantly for your argument, the FDA labeling says the device should not be used in people receiving calcium channel blockers or agents blocking L-type or T-type voltage-gated calcium channels unless treatment is modified.
Mechanistically, the best-supported tumor-channel paper behind that platform points to CACNA1H / CaV3.2, not CaV1.4. The 2019 EBioMedicine study reported that tumor-specific amplitude-modulated RF EMF in hepatocellular carcinoma acts through Ca2+ influx via Cav3.2 T-type channels, and the search-indexed abstract states that this calcium influx is the necessary and sufficient upstream event for the anti-proliferative effect in HCC cells. So P1 does not prove the same channel mechanism as the Cell paper. What it does prove is that a clinically used, non-thermal EMF platform can depend on voltage-gated calcium-channel biology strongly enough for both the published mechanism and FDA labeling to reflect it.
Put the two together, and the scientific conclusion becomes sharper. The Cell paper gives one controlled ELF-like burst regime linked to CYB5B plus CaV1.4/CACNA1F. TheraBionic gives a clinically deployed AM RF regime linked to CaV3.2/CACNA1H. Different channel subtypes. Different frequency architecture. Same broad lesson: field structure can matter biologically through calcium-dependent pathways even when the effect is not being explained by heating. That is the claim worth making.
This is exactly where your uploaded “S4-Mito-Spin,” “bioelectric dissonance,” and “ceLLM” framework is strongest. Its best claim is not that EMF has already been proven to cause every downstream disease you diagrammed. Its best claim is that cells are timing-sensitive systems, and that non-native EMF might act not only as an energy exposure but as an information-quality problem — a perturbation of signal fidelity upstream of many different cellular outputs.
Biologically, that is a plausible hypothesis. Pancreatic islets use coherent electrical and calcium oscillations to drive pulsatile insulin secretion. T cells and other immune cells decode calcium timing through calcineurin/NFAT and related pathways. More generally, calcium signaling is known to carry information through frequency modulation and temporal patterning. Once that is true, the upstream question changes. The issue is no longer only whether a field raises calcium. The issue becomes whether a field writes a clean, decodable rhythm or injects timing noise into a system that depends on temporal precision.
That is the core idea your image set is visualizing. A clean periodic field, in a permissive biological context, could in principle create an orderly calcium waveform that the cell can interpret. A mismatched or irregular field could instead create three kinds of error: phase error (the signal arrives at the wrong biological moment), duty-cycle error (the on/off structure is wrong), and coherence loss (neighboring cells stop oscillating together). In your terms, that is the transition from high-fidelity signaling to bioelectric dissonance.
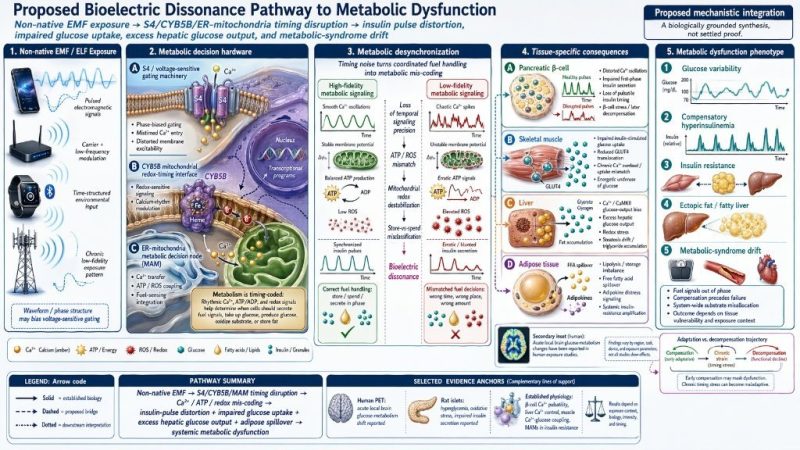
Seen through that lens, your metabolic dysfunction panel is one of the more grounded figures. Beta cells and islets genuinely use oscillatory calcium dynamics to coordinate insulin release, so a hypothesis in which timing noise distorts insulin pulsatility, mitochondrial ATP/ROS coupling, GLUT4 recruitment, or hepatic fuel handling is physiologically plausible. The missing piece is not the plausibility of the timing code; it is direct proof that realistic non-therapeutic EMF exposures corrupt that code in vivo under blinded, dosimetry-controlled conditions.

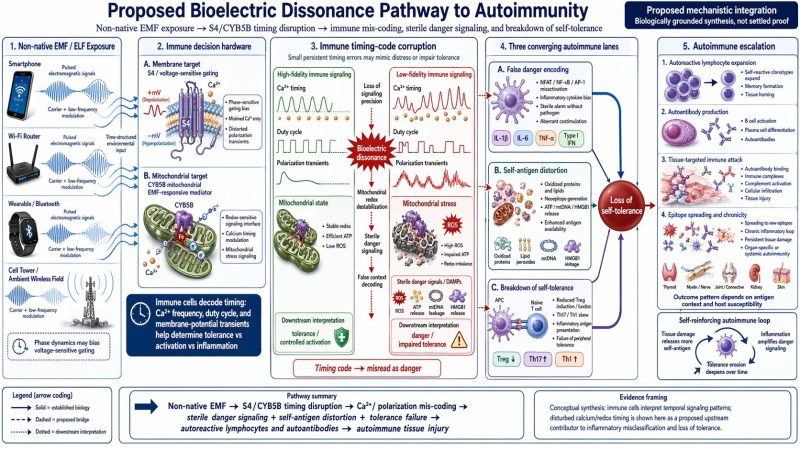
The autoimmunity panel also has a legitimate upstream logic. Immune activation is not just a matter of calcium flood; it is shaped by calcium timing and by the downstream behavior of calcineurin/NFAT, NF-κB, AP-1, and related transcriptional modules. That makes a timing-error hypothesis reasonable at the signaling level. But the longer chain from exposure to sterile danger encoding, failed self-tolerance, autoantibodies, and organ-specific autoimmunity is still far beyond what current EMF studies have directly demonstrated. It belongs in a blog as a proposed mechanistic branch, not as a concluded disease pathway.
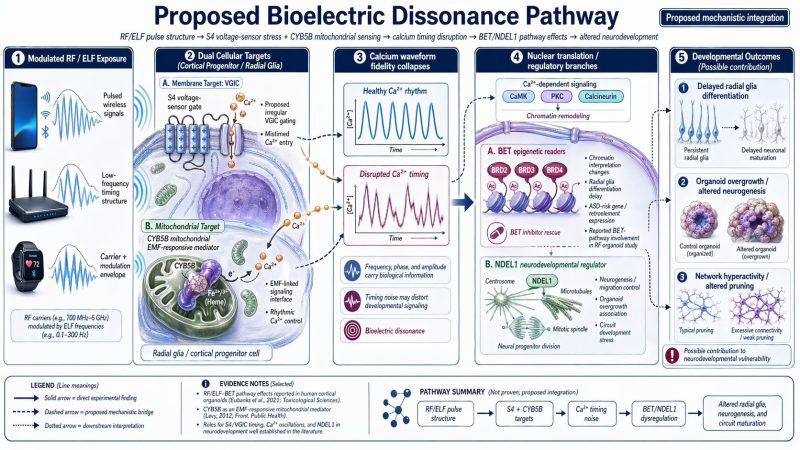
The neurodevelopment panel is the strongest of the disease-specific figures because it overlaps with actual recent organoid data. A 2025 Cell Reports paper reported that RF exposure altered radial glia differentiation in human cortical organoids, maintained stem-cell identity, and induced ASD-related gene and retroelement expression, with BET-mediated pathways prominently involved. Separately, a 2024 autism organoid study reported that Ndel1 activity was highly correlated with organoid growth rate and size. That does not prove your full proposed chain of S4/CYB5B → calcium timing noise → BET/NDEL1 dysregulation, but it does mean that this branch of the model is not pure speculation. There is already experimental pressure on the downstream side of that map.
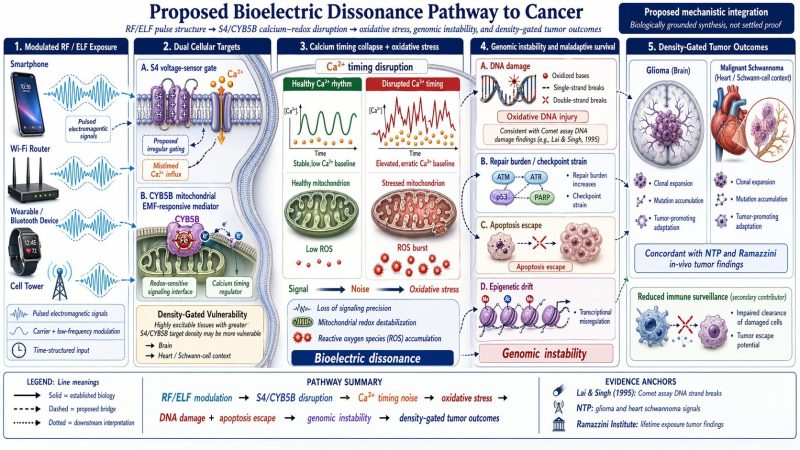
The cancer panel sits in the middle: more grounded than the ceLLM backpropagation figure, but still not mechanistically closed. NTP reports that high exposure to cell-phone-type RFR in male rats was associated with malignant schwannomas in the heart and some evidence of malignant gliomas in the brain, and a 2025 systematic review of animal cancer studies concluded that the certainty of evidence was strongest for malignant heart schwannomas and gliomas. That supports keeping the cancer schematic in the conversation. It does not yet prove your specific upstream S4/CYB5B-to-genomic-instability route for real-world human exposure scenarios.

Where the image set becomes much more speculative is in the last three conceptual panels. The Cellular Goldilocks Zone works as a framing metaphor about nested environmental conditions.
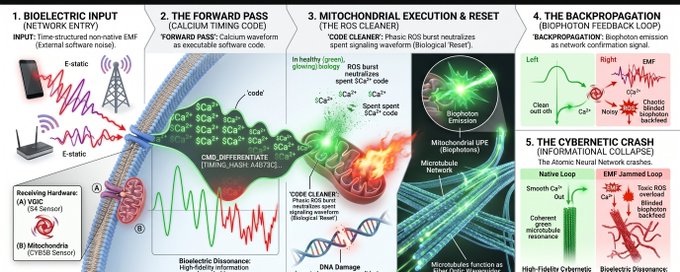
The Weak Magnetic Co-Zeitgeber panel is an interesting chronobiology hypothesis, but should stay labeled as exploratory. The Geometry of Evolution and ceLLM / backpropagation graphics are the boldest leap: they are useful as research-program language or systems metaphors, but they should not be presented as if mainstream cell biology has already accepted DNA topology as a literal neural-network weight matrix or mitochondria as a proven backpropagation layer. Those panels are strongest when they are clearly marked as a conceptual synthesis, not a settled mechanism.
That distinction matters because the current mainstream evidence base is still more conservative than your model. SCHEER has said it could not identify moderate or strong evidence for adverse health effects from chronic or acute RF-EMF exposure below current European limits, and the 2024 WHO-commissioned human observational review concluded that RF from mobile phone use likely does not increase the risk of brain cancer. So the honest posture is not that your framework has already won the argument. It is that the old thermal-only dismissal is now too simplistic, while the generalized disease claims still require much tighter experimental proof.
That, in my view, is the blog-worthy message. The Cell paper shows that a defined EMF program can be converted into a CYB5B-linked, calcium-oscillation-based gene-control signal. TheraBionic P1 shows that a non-thermal, amplitude-modulated RF platform can be clinically relevant and calcium-channel-dependent. Your framework then asks the right next question: when does an external field write a functional rhythm, and when does it inject biological static? That question is no longer fringe. It is not settled, but it is now specific enough that nobody serious should brush it away with “it doesn’t heat tissue, therefore nothing is happening.”







